Scalp seborrheic dermatitis (SD) is a chronic type of inflammatory dermatosis associated with the proliferation of Malassezia species. Several recent scalp microbiome studies on different populations have also revealed the association of dandruff with bacterial and fungal dysbiosis. Cutibacterium acnes (formerly known as Propionibacterium acnes) and Staphylococcus epidermidis were noted to be the core bacterial species on the top of the scalp.
ABSTRACT
Background
Scalp seborrheic dermatitis (SD) is a chronic inflammatory dermatosis associated with sebum imbalance and proliferation of Malassezia species. Various antifungal shampoos are commonly used for scalp SD.
Aims
Glycyrrhetinic acid is known to have antioxidative, anti-inflammatory, and anti-allergic effects. This study was designed to evaluate the effectiveness of a new-formula shampoo that contains glycyrrhetinic acid for the treatment of scalp SD.
Patients/Methods
Thirty-four patients were enrolled and treated with the 6% glycyrrhetinic acid complex shampoo. Efficacy was assessed clinically with Dermatology Life Quality Index (DLQI) and Adherent Scalp Flaking Score (ASFS) by the same dermatologist at baseline, week 2, and week 5. Among the 24 subjects with the most significant clinical improvement, four common microorganisms from scalp samples were analyzed by quantitative polymerase chain reaction (qPCR) at baseline, and week .
Results
The DLQI and ASFS at week 2 and week 5 improved significantly relative to baseline. The bacteria profiles showed a significant increase of Cutibacterium acnes and a decrease of Staphylococcus epidermidis at week 5. The fungi profiles showed significant decreases of both Malassezia restricta and Malassezia globosa. The ratio of C. acne to S. epidermidis increased significantly from 0.93 at baseline to 1.55 at week 5. The ratio of M. restricta to M. globosa decreased from 5.02 at baseline to 1.00 at week 5. Conclusions
The effectiveness of this new regimen was objectively demonstrated at the clinical and microbiological levels. This new formula may alleviate the bacterial and fungal dysbiosis in scalp SD.
INTRODUCTION
Scalp seborrheic dermatitis (SD) is a chronic type of inflammatory dermatosis associated with the proliferation of Malassezia species.1, 2 Several recent scalp microbiome studies on different populations have also revealed the association of dandruff with bacterial and fungal dysbiosis.3 Cutibacterium acnes (formerly known as Propionibacterium acnes) and Staphylococcus epidermidis were noted to be the core bacterial species on the top of the scalp. The former was associated with a healthier scalp, and the latter with a dandruff scalp.2 Along with the common Malassezia species (M. restricta and M. globosa) on the scalp, a high association of dandruff with other Malassezia species was also observed in the mycobiome.2, 4
Antifungal therapies against Malassezia species and anti-inflammation agents are helpful for scalp SD. Traditionally, ketoconazole, ciclopirox olamine, zinc-pyrithione, and selenium sulfide shampoos are common treatments for this condition.5 However, the treatment response rates varied from 26% to 77%.6 A new shampoo with different compounds is still needed for patients with a poor response to these conventional shampoos.
Glycyrrhetinic acid, the main metabolite of glycyrrhizin from licorice root, is known to have antioxidative, anti-inflammatory, anti-allergic, and antimicrobial effects. It can be used for various skin conditions including pruritus, atopic dermatitis, acne vulgaris, skin aging, and hyperpigmentation.7 Its application for scalp SD was rarely found in the literature.8 This open-labeled pilot study aims to evaluate the clinical efficacy of a new-formula shampoo containing glycyrrhetinic acid. The microorganisms commonly found on the scalp were analyzed to examine a possible dysbiosis restoration in scalp SD patients.
MATERIALS AND METHODS
2.1 Patients
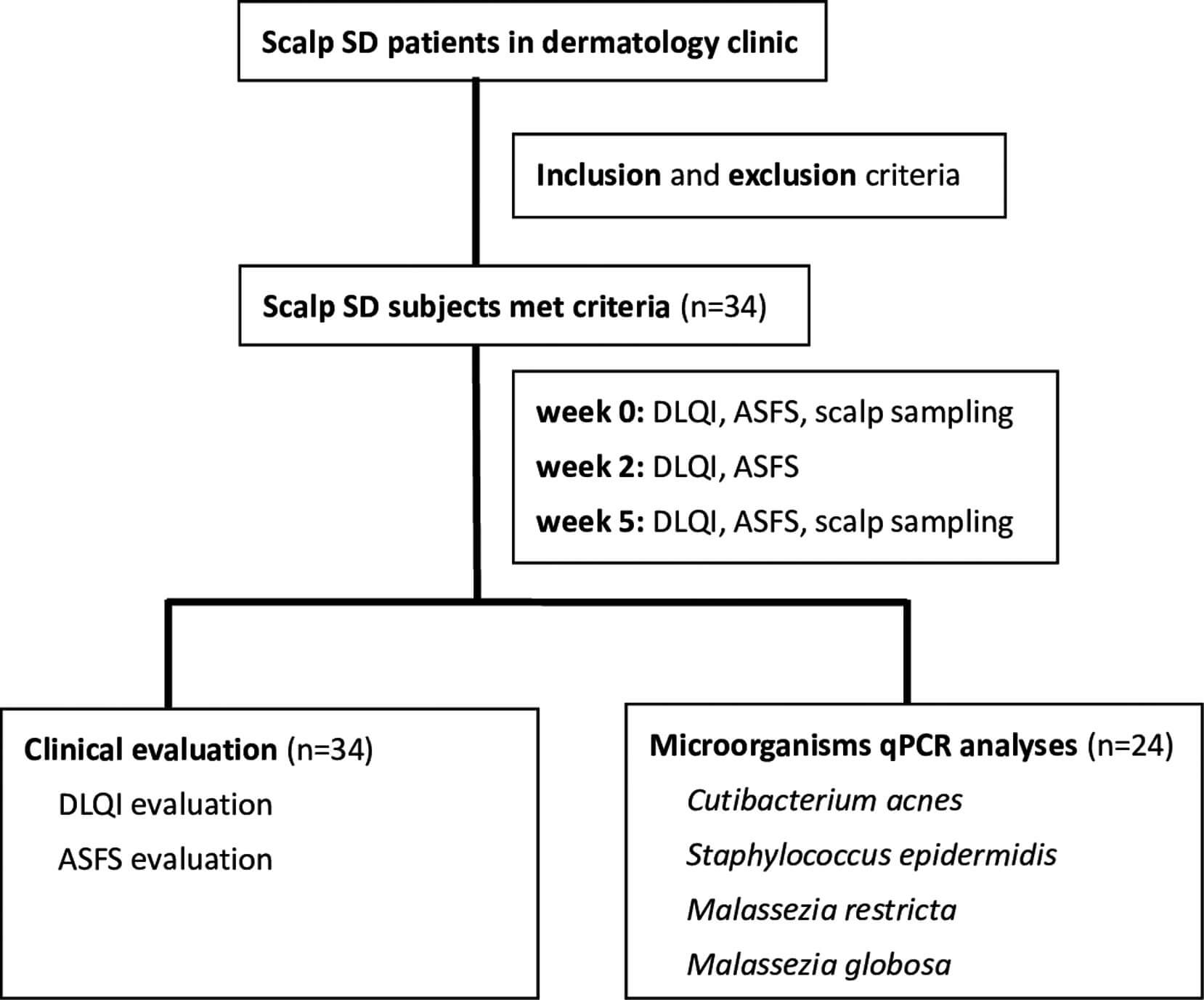
Patients aged 20–65 years who visited En Chu Kong Hospital for scalp SD from June 2020 to February 2021 were enrolled in this study with inclusion criteria of Dermatology Life Quality Index (DLQI) above 2 and Adherent Scalp Flaking Score (ASFS) above 10.9–11 The purpose of this non-invasive study was open for all patients, and informed consent forms were signed (IRB No. ECKIRB1090405). Patients were excluded from the study if they were allergic to menthol, had scalp surgery in the previous month, planned to have pregnancy or breastfeeding, had autoimmune, cancer, renal, or liver diseases, or had any other major systemic diseases. In total, 34 patients met the inclusion criteria. The study algorithm was shown in Figure 1.
FIGURE 1: Study algorithm. SD, seborrheic dermatitis; DLQI, Dermatology Life Quality Index; ASFS, Adherent Scalp Flaking Score; qPCR, quantitative polymerase chain reaction
2.2 Test products
The ingredients of the new-formula shampoo with 6% glycyrrhetinic acid complex (5α Juniper MediPRO, MacroHi Co. Ltd.) are shown in Table 1. All patients were instructed to rinse and massage their scalps first with the assigned shampoo for 10–20 s and rinse off with water, followed by a second massage for 1–2 min with the same product and rinse off everyday for 5 weeks. Other drugs or external preparations for scalp diseases were not allowed during the study period.
2.3 Clinical evaluation
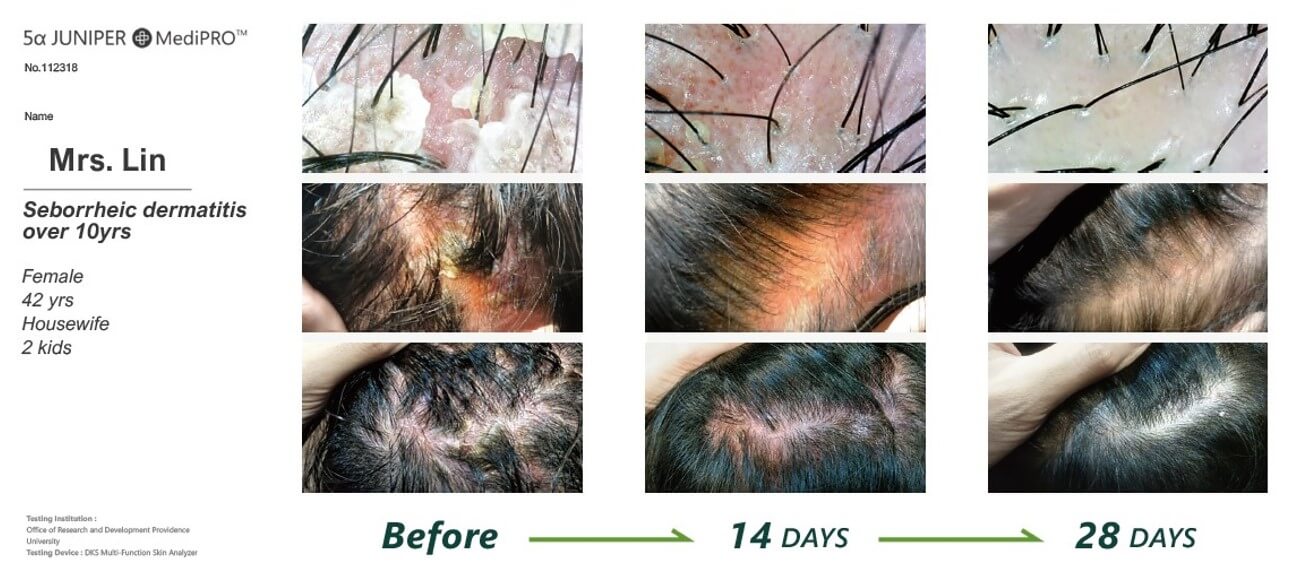
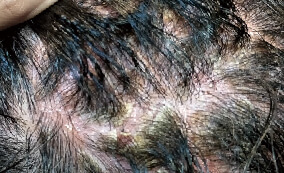
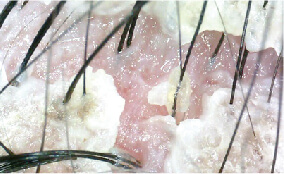
One of the study cases: Seborrheic dermatitis over 10 years
Female/ 42 years old
Housewife with 2 kids

Pictures before the MediPRO treatment
2.4 Sample collection
Four common microorganisms, namely, C. acnes, S. epidermidis, M. restricta, and M. globosa, were sampled from scalp and analyzed by quantitative polymerase chain reaction (qPCR) at baseline and week 5. Patients were asked not to wash their hair on the day before the sampling procedure. A controlled environment with a room temperature of 25 ± 2°C and a 50% ± 10% relative humidity was provided during scalp sampling. The sample was collected from the vertex region and temporal region above the ears in triplicate. Sampling was conducted by the swab method as described in previous studies.12
Presto™ Mini gDNA Bacteria Kit (GBB100, Geneaid) was used as per the manufacturer’s instructions for the preparation of the samples. Sterile swabs (LIBO Medical Products Inc., Cat. No. 30071) were soaked in SCF-1 solution (pH 7.6 Tris buffer 50 mM, pH 8.0 EDTA 1 mM, and 0.5% Tween 20) before sampling. A comb was used to separate hair fibers, and the pre-wetted swab was rubbed back and forth onto the scalp surface for about 30 times. The total covering surface was about 5 cm2 (10 cm in length and 0.5 cm in width). After sampling, the head of each swab was cut from the handle and placed into a tube containing 1.5 mL SCF-1 solution. All samples were stored at −20°C until DNA extraction.
2.5 Bacterial and fungal genomic DNA extraction
Among the 24 subjects with the most significant clinical improvement, genomic DNA was extracted from the swab samples using DNeasy® Blood & Tissue Kit (Qiagen, Cat. No. 69504) according to the manufacturer’s instructions. The procedure was slightly modified to extract fungal genomic DNA.
The swab samples were vortex mixed at the maximum speed for 30 s, and the swab heads were then removed. Two identical 1.5 mL suspensions were generated from duplicate collection tubes. One was for bacterial DNA, and the other was for fungal DNA extraction. The suspension from each tube was pelleted by centrifugation at 20 000 × g (14 000 rpm) for 3–5 min.
For bacterial DNA extraction, the manufacturer’s protocol for genomic DNA isolation from Gram-positive bacteria was followed. For fungal DNA extraction, the protocol with minor modifications was used: The pellet was re-suspended in 150 μL of lysis buffer (200 mM Tris-HCl, pH 8.0; 25 mM EDTA, 250 mM NaCl, and 0.5% SDS) at 100°C for 15 min. Later, 180 μL of buffer ATL was added and incubated with 20 μL of proteinase K at 56°C for 15 min. The remaining steps were performed according to the manufacturer’s protocol, and samples were eluted in 150 μL AE buffer. The extracted DNA was stored at −20°C before real-time PCR quantification.
2.6 Real-time quantitative PCR
The four common species, namely, C. acnes, S. epidermidis, M. restricta, and M. globosa, were collected from the scalp and quantified with real-time PCR using TaqMan probes and specific primers listed in Table 2. Real-time quantitative PCR was performed by StepOnePlus™ System (Applied Biosystems) with optical 96-well plates. The reaction mix had a total volume of 15 μL and consisted of 1× KAPA PROBE FAST qPCR Master Mix (Kapa Biosystems), 250 nM forward and reverse primers, 300 nM probe, and 5 μL sample. The reaction conditions were 95°C for 3 min, 40 cycles of 95°C for 3 s, and 60°C for 1 min. Each sample was run in triplicate. The copy numbers of each sample were calculated from the standard curves as follows.
| Species | Primer | Sequence (5′–3′) |
|---|---|---|
| Cutibacterium acnes | PA-F | GCGTGAGTGACGGTAATGGGTA |
| PA-R | TTCCGACGCGATCAACC | |
| Staphylococcus epidermidis | SepV58 | GCTGTGATGGGGAGAGGAAAT |
| SepR54bSta59bT | CGGTACGGGCACCTGTTATC | |
| Malassezia restricta | qMA-F | GTGAATTGCAGAATTCCGTGAAT |
| qMR-R | GCGAGCCTGTGCTAGGTA | |
| Malassezia globosa | qMA-F | GTGAATTGCAGAATTCCGTGAAT |
| qMG-R | GAGCTTTTTCTAGAGAAGAAAAG |
2.7 Standards curve for quantification
The genomic DNA of C. acnes (ATCC 29399), S. epidermidis (ATCC 14990), M. restricta (ATCC MYA-4611), and M. globosa (ATCC 96807) was extracted as mentioned previously. The targeted fragments detected by primers and probes listed in Table 2 were amplified using the new primer sets. The new primer sets designed by using Primer3web (http://primer3.ut.ee/) are listed in Table 3. The amplified products were purified by GenepHlow™ Gel/PCR Kit (Geneaid, Cat. No. DFH100) and cloned into pGEM®-T Easy Vector (Promega, Cat. No. A1360), followed by transformation into DH5α competent E. coli cells. Plasmids were extracted using a High-Speed Plasmid Mini Kit (Geneaid, Cat. No. PD300), and the concentrations of the products were determined by Nanodrop 1000 Spectrophotometer (Thermo Fisher Scientific Instrument Co. Ltd). The standard curves were generated using a tenfold dilution of the four plasmids separately. Serially diluted plasmids in ddH2O to a final concentration ranging from 101 to 106 copies/ml were used for real-time PCR. A new standard curve was run for each real-time PCR reaction.
Sequences of designed primer sets for four microorganism species
| Species | Primer | Sequence (5′–3′) |
|---|---|---|
| Cutibacterium acnes | CA-F | GCGTGAGTGACGGTAATGGGTA |
| CA-R | TTCCGACGCGATCAACC | |
| CA-TAQ | AGCGTTGTCCGGATTTATTGGGCG | |
| Staphylococcus epidermidis | SE-F | GCTGTGATGGGGAGAGGAAAT |
| SE-R | CGGTACGGGCACCTGTTATC | |
| SE-TAQ | AGAGGCTTTTCTCGGCAGTGTGAAATCAACGA | |
| Malassezia restricta | MR-F | GTGAATTGCAGAATTCCGTGAAT |
| MR-R | GCGAGCCTGTGCTAGGTA | |
| MR-TAQ | CTTTGAACGCACCTTGCGCTC | |
| Malassezia globosa | MG-F | GTGAATTGCAGAATTCCGTGAAT |
| MG-R | GAGCTTTTTCTAGAGAAGAAAAG | |
| MG-TAQ | CTTTGAACGCACCTTGCGCTC |
2.8 Statistical analysis
The therapeutic efficacy analysis of the DLQI and ASFS score during the treatment phase was performed by comparison of mean ± standard deviation (SD) at week 2 and week 5 versus baseline with Scheffé Test, respectively. The log copy numbers of 4 microorganisms at baseline and at week 5 were compared by a paired sample t-test. SPSS software 26.0 was used for analysis. All statistical tests were set at a significance level of 0.05.
RESULTS
3.1 Changes in clinical severity score
A total of 34 patients, 12 males and 22 females, completed the clinical study. The clinical severity score DLQI and ASFS at baseline and after treatment are listed in Tables 4 and 5. The DLQI and ASFS improved significantly relative to baseline at weeks 2 and week 5, respectively (p < 0.05). However, the changes in DLQI and ASFS between weeks 2 and week 5 were not significant (p = 0.652).
One of the study cases: Seborrheic dermatitis over 10 years
Female/ 42 years old
Housewife with 2 kids

Pictures AFTER 28 days with MediPRO treatment
| Clinical parameter | DLQI (mean ± SD) |
|---|---|
| Week 0 | 12.7 ± 5.2 |
| Week 2 | 6.5 ± 5.7* |
| Week 5 | 5.3 ± 5.9** |
| % change W2 vs. W0 | −53.8 |
| % change W5 vs. W0 | −54.3 |
- Abbreviations: DLQI, Dermatology Life Quality Index; SD, standard deviation; W0, week 0; W2, week 2; W5, week 5.
- * Comparison of mean ± standard deviation (SD) change versus W0 using Scheffé Test, p < 0.05.
- ** Comparison of mean ± standard deviation (SD) change versus W0 using Scheffé Test, p < 0.05.
| Clinical parameter | ASFS (mean ± SD) |
|---|---|
| Week 0 | 23.1 ± 8.9 |
| Week 2 | 10.4 ± 10.7* |
| Week 5 | 9.1 ± 13.0** |
| % change W2 vs. W0 | −56.9 |
| % change W5 vs. W0 | −65.8 |
- Abbreviations: ASFS, Adherent Scalp Flaking Score; SD, standard deviation; W0, week 0; W2, week 2; W5, week 5.
- * Comparison of mean ± standard deviation (SD) change versus W0 using Scheffé Test, p < 0.05.
- ** Comparison of mean ± standard deviation (SD) change versus W0 using Scheffé Test, p < 0.05.
3.2 Changes in four common microorganisms
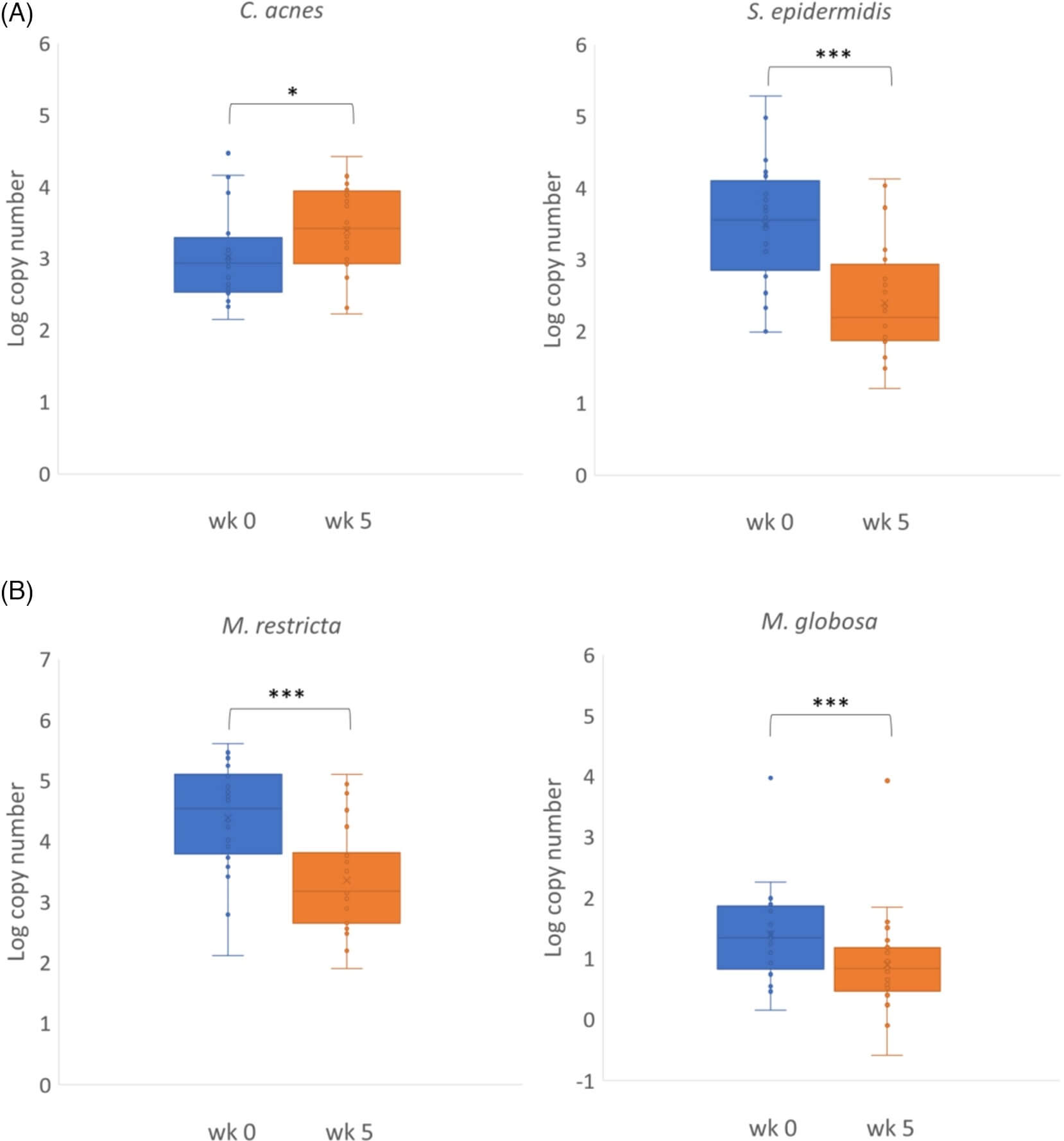
Among the 24 subjects with the most significant clinical improvement, the amount of the four common microorganisms on the scalp showed significant changes after 5 weeks of glycyrrhetinic acid shampoo treatment (Figure 2). The bacteria profiles showed a significant increase of C. acnes (p < 0.05) and a significant decrease of S. epidermidis (p < 0.001), respectively (Figure 2A). On the contrary, the fungi profiles showed a significant decrease of both M. restricta (p < 0.001) and M. globosa (p < 0.001) (Figure 2B).
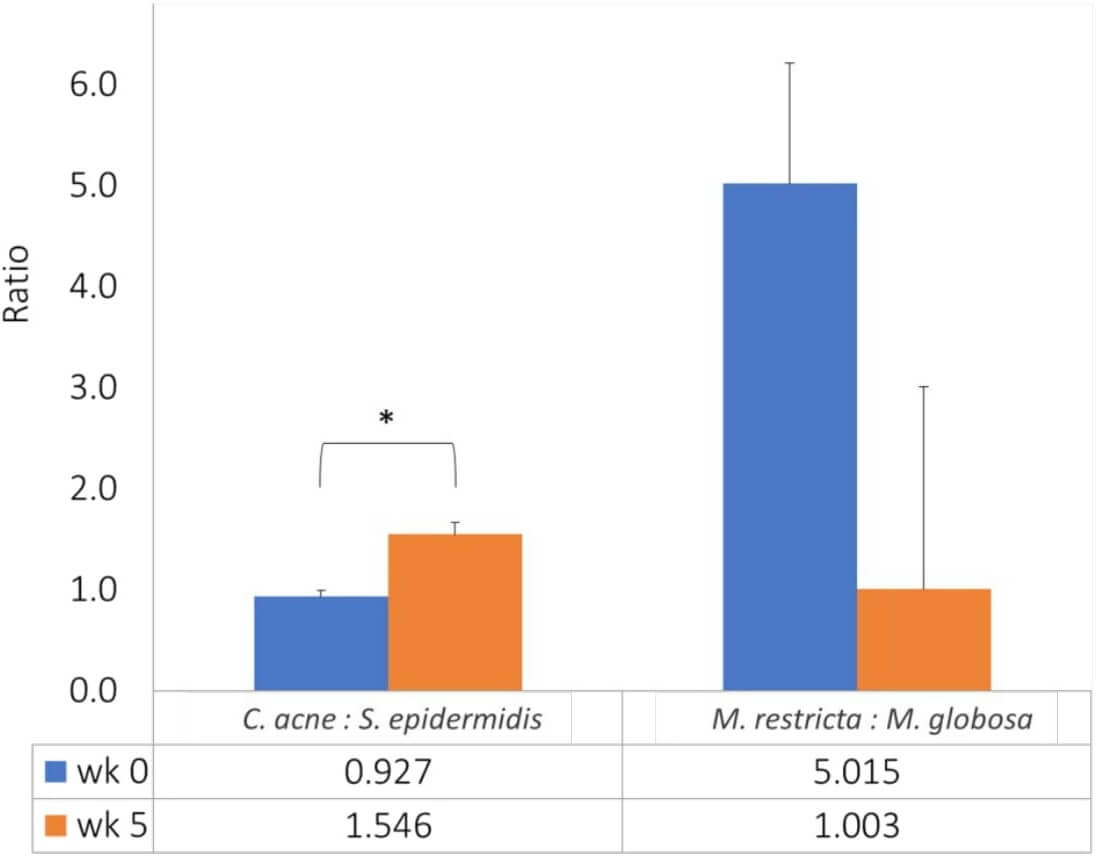
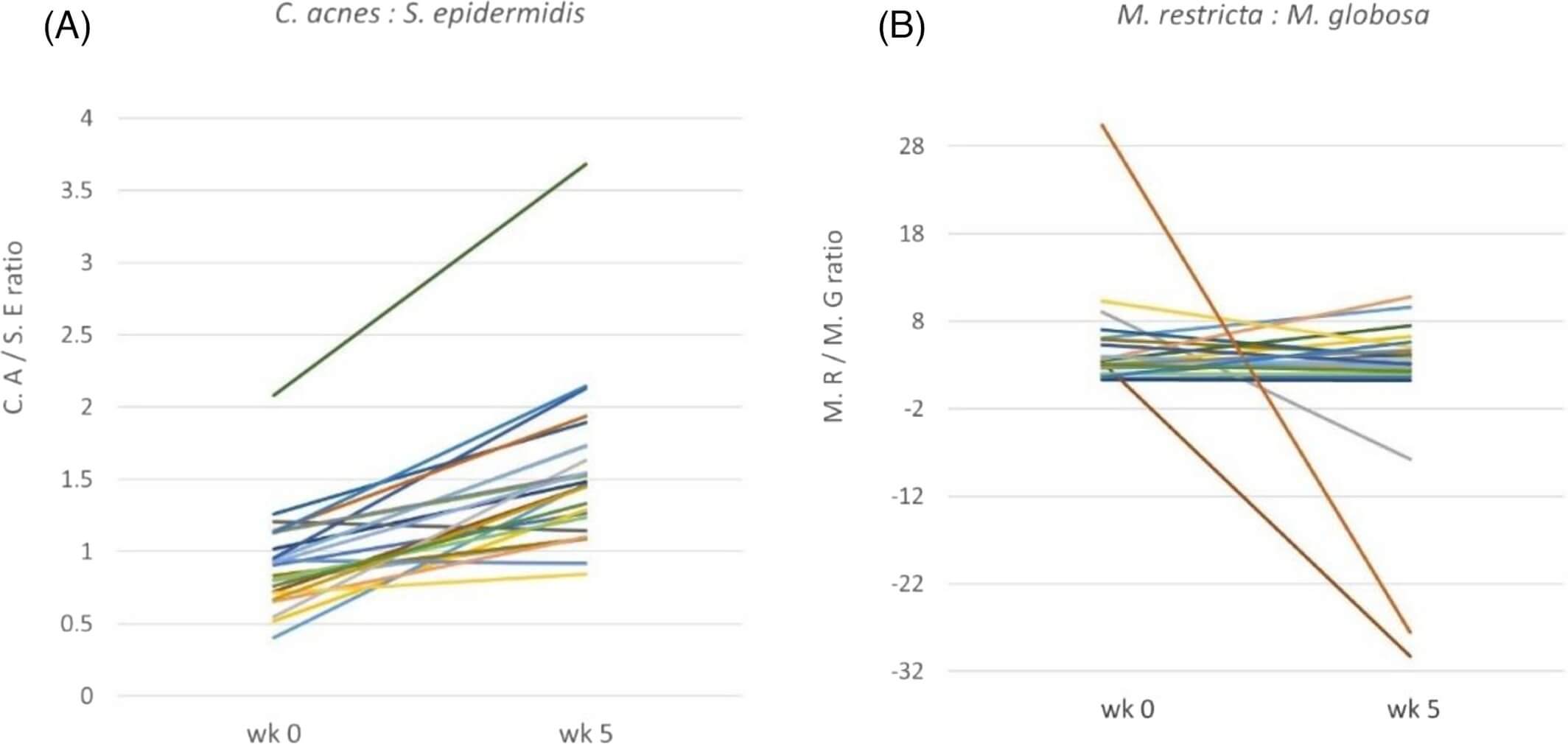
We further analyzed the changes in the ratio of C. acne to S. epidermidis and the ratio of M. restricta to M. globosa from week 0 to week 5. The ratio of C. acne to S. epidermidis increased significantly from 0.93 at baseline to 1.55 at week 5 (p < 0.001). In contrast, the ratio of M. restricta to M. globosa decreased from 5.02 at baseline to 1.00 at week 5, but the difference was not statistically significant (p = 0.176) (Figure 3). The 24 individual changes of the bacteria ratio (C. acne to S. epidermidis) showed increases in 22 subjects after a 5-week intervention of glycyrrhetinic acid shampoo (Figure 4A). The individual changes of the fungi ratio (M. restricta to M. globosa) showed variable trends from week 0 to week 5 (Figure 4B). Among these 24 subjects, 13 patients had decreasing trends after 5 weeks.
DISCUSSION
The pathogenesis of SD is complex and was hypothesized to result from the interactions among cutaneous microflora, the immune system, and the sebum imbalance in the stratum corneum of the scalp.13 It is known to be a chronic type of inflammatory dermatosis associated with the proliferation of Malassezia species.1, 2 Traditionally, topical steroids have been used to reduce scalp inflammation. However, Malassezia species cannot be inhibited by steroids. In addition, the chronic nature of the disease and the need for maintenance therapy should be considered, a long-term treatment is inevitable. It is obvious that steroids are not appropriate for long-term and prophylactic use owing to the relapsing nature of SD.
Glycyrrhetinic acid and its derivatives showed a remarkably antimicrobial effect with its minimum inhibitory concentration (MIC) value against S. epidermidis at 12.5 ug/ml.7 Our results showed a significant decrease of scalp S. epidermidis after a 5-week use with glycyrrhetinic acid shampoo. This ingredient also helped to significantly increase C. acnes accordingly. The overall ratio of C. acne to S. epidermidis increased significantly from 0.93 at baseline to 1.55 after a 5-week treatment. We also found that 22 of 24 individuals showed similar increasing trends of the bacteria ratio (C. acne to S. epidermidis). These phenomena proved that the overall increase of C. acne may be attributed to the reciprocal inhibition of these two bacteria. Also, the increased ratio of C. acne to S. epidermidis implies a healthier scalp condition achieved after the glycyrrhetinic acid intervention. This new formula seemed to be an effective way to adjust the balance of these two dominant bacteria on the scalp by the enhancement of Cutibacterium and the suppression of Staphylococcus.
Although glycyrrhetinic acid alone has been found to have a weaker effect against fungi, our results showed a significant reduction (p < 0.001) in the number of M. restricta and M. globosa after 5 weeks of glycyrrhetinic acid intervention. According to Saxena et al., a lower M. restricta to M. globosa ratio was thought to be associated with a healthier scalp.4 The overall ratio of M. restricta to M. globosa decreased from 5.02 at baseline to 1.00 at week 5 in our study, but the result was not statistically significant. The insignificant change may be due to the variable trends among the 24 individual fungi ratios (M. restricta to M. globosa) and the small sample size of this research. The clinical significance of this ratio and its application in scalp SD severity assessment may need further research to validate this concept. Our result also indicated that bacteria imbalance might have a more substantial impact on the severity of dandruff than fungi. A larger controlled study will be required to give better statistical data and reveal the complex interactions among the bacteria, fungi, sebum, and hydration of the scalp SD.
In conclusion, our research showed a significant treatment response for scalp SD by 6% glycyrrhetinic acid complex shampoo. The most important role of glycyrrhetinic acid is its anti-inflammatory effect and its substantial antimicrobial activity toward some strains of bacteria and fungi. The effectiveness of this new regimen was objectively demonstrated with DLQI and ASFS clinically as early as 2 weeks after treatment. The inhibition of S. epidermidis as well as Malassezia species alleviated the dysbiosis and restored microbiota. Since these beneficial effects might be attributed to the detergents or other ancillary ingredients in this product, further randomized double-blinded placebo-controlled studies are mandatory in the future.
4.1 Limitation of Study
The present study did not represent the actual daily scenario because it was designed to use shampoo every day for 5 weeks. The duration of the study was short, and the sample size was small. This non-randomized, open-label research also lack a control group for comparison. The analyses of 4 microorganisms were only performed in 24 out of the 34 subjects due to a limited budget. The long-term results of the product could not be assessed. Finally, the beneficial effects might be attributed to the detergents or other ancillary ingredients in this product. Further randomized double-blinded placebo-controlled studies are mandatory in the future.
ACKNOWLEDGEMENTS
The authors would like to thank Wan-Hsuan Lee and Zih-Han Hsu for their assistance on the scalp examination process and Pei-Yi Xu for the participant management and recruitment.
CONFLICT OF INTEREST
This study was supported by MacroHi Co. Ltd., Taiwan. CS Wang and HC Wang received fees for their roles as investigators in this study. HH Chen received fees for manuscript preparation.
AUTHOR CONTRIBUTIONS
HC Wang and CS Wang designed the study and performed clinical assessments. SC Hsieh and YT Hung supervised the sequencing experiments and subsequent analyses of microorganisms. HC Wang, CS Wang, and HH Chen analyzed the data. HH Chen wrote the manuscript. HC Wang, CS Wang, and SC Hsieh reviewed the manuscript. The final manuscript was read and approved by all authors. ETHICAL APPROVAL
All procedures were in accordance with legal requirements in the country and the ethical principles stated in the Declaration of Helsinki of 1975, as revised in 2000 and 2008. The study was approved by the En Chu Kong Hospital Ethics Committee (IRB No. ECKIRB1090405). All patients signed the informed consent forms before the study. REFERENCE